|
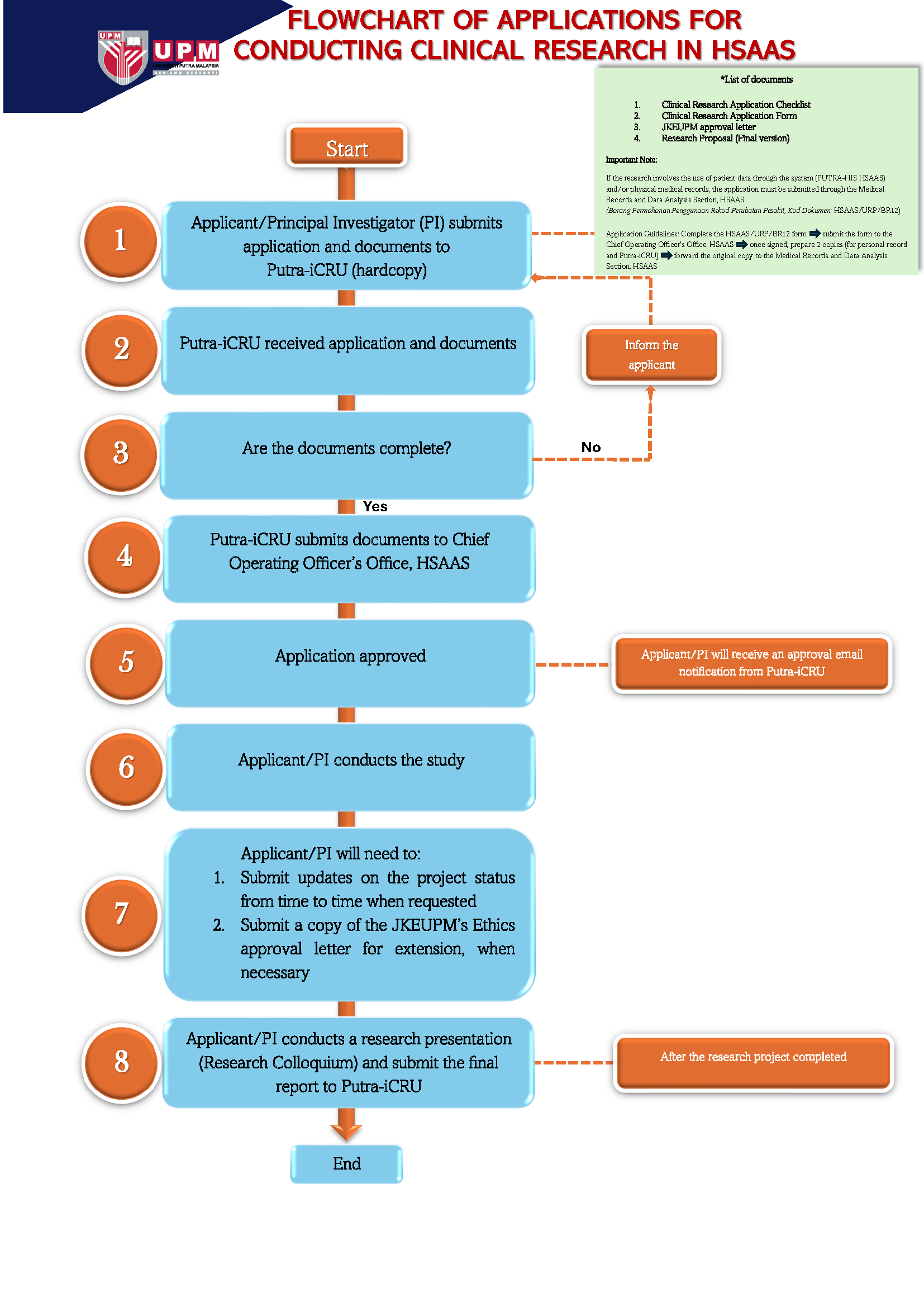
Researcher who want make application to conduct research at Hospital Sultan Abdul Aziz Shah (HSAAS) must prepared research document and submit the document (as a hardcopy) to Putra-iCRU, HSAAS for review and further action.
- Researchers need to download the following documents and attach supporting documents:
Document:
- Senarai Semak Permohonan Penyelidikan Klinikal

- Clinical Research Application Form

- Letter of JKEUPM Approval (Ethic Committee For Research Involving Human Subject)
- Research Proposal (FINAL)
- If the research project is managed by UPMCS, please attach a copy of the agreement document (CTA/MOA/CDA/NDA)
- Every clinical research application at HSAAS must obtain approval from the head of department for each facility used to conduct research. For reference: List of Head Department
- Request cooperation researchers to ensure:
- If the researcher's application is from outside the HSAAS, please obtain the Site Investigator (HSAAS internal researcher) as a co-investigator before submitting the research application document to the Putra-iCRU.
For the purpose of JKEUPM Approval (Ethic Committee For Research Involving Human Subject). Please refer to the JKEUPM secretariat at the following link: JKEUPM
Reference for JKEUPM Approval process: JKEUPM Approval
Garis Panduan Pengurusan Penyelidikan Klinikal : GPK
|
|
FLOWCHART OF APPLICATIONS FOR CONDUCTING RESEARCH WITHOUT AGREEMENT IN
HOSPITAL SULTAN ABDUL AZIZ SHAH (HSAAS)
TERMINOLOGY AND GLOSSARY:
Putra-iCRU - Putra Integrated Clinical Research Unit
JKEUPM - Jawatankuasa Etika Universiti Putra Malaysia
|
Updated:: 13/04/2026 [intanbasirah]
MEDIA SHARING