|
Researcher who want conduct Clinical Trial research at HPUPM must make application to Putra-iCRU with prepared research document and submit the document (as a hardcopy) to Putra-iCRU office, for review and further action.

*Each application for clinical research at HSAAS must obtain approval from the head of department for each facility used*
*If the application is from outside HSAAS, please get the Site Investigator first before sending the research application to Putra-iCRU*
*Pemakluman Berkenaan Caj Yuran Pengurusan (Management Fees) 15% bagi Semua Terimaan Geran Projek Penyelidikan Klinikal*
Document:
- Senarai Semak Permohonan Penyelidikan Klinikal (as a hardcopy)

- Clinical Research Application Form (as a hardcopy)

- Letter of Approval JKEUPM (Ethics Committee for Research Involving Human Subjects) (as a hardcopy)
- MOA Application Form (online)

- Template Implikasi Kewangan_Pelaporan JKTK_UPM_2025 (online)

- Pemakluman Peratusan Caj Pengurusan Geran Penyelidikan Klinikal

- TOR of Clinical Trial Agreement (CTA) in HSAAS

- Application Form for Opening of Trust Account (as a hardcopy)

- Non-Participation Reply Form (NPRF)

- If the research project is managed by UPMCS, please attach a copy of the agreement document (CTA/MOA/CDA/NDA)
- Every clinical research application at HSAAS must obtain approval from the head of department for each facility used to conduct research
Garis Panduan Pengurusan Penyelidikan Klinikal : GPK
|
|
|
|
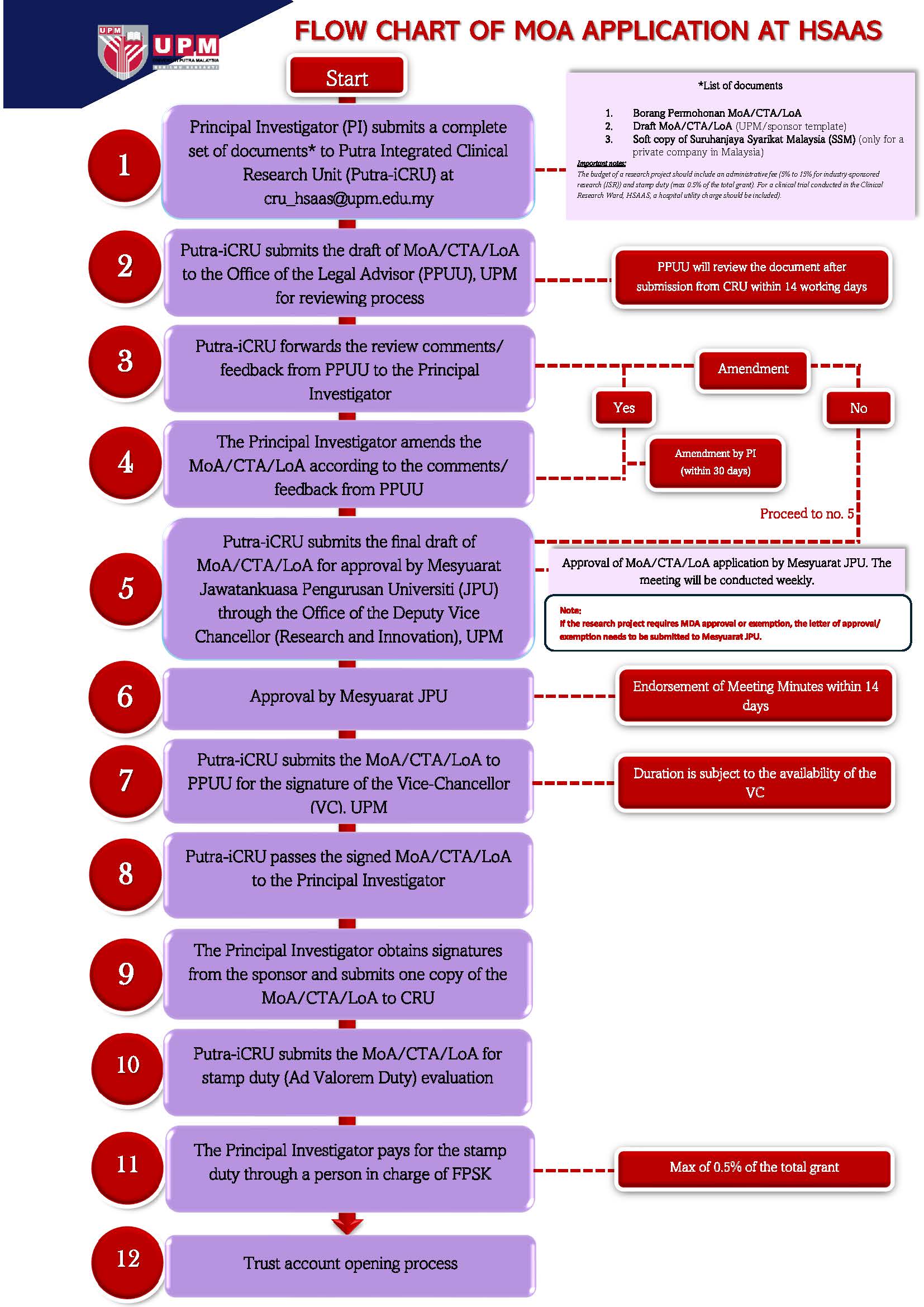
FLOWCHART OF MOA APPLICATION AT
HOSPITAL SULTAN ABDUL AZIZ SHAH (HSAAS)
|
|
Updated:: 13/04/2026 [intanbasirah]
MEDIA SHARING